CDSCO Releases Draft Guidance To Align New Drugs And Clinical Trials Rules And SUGAM For Stakeholder’s Comments
New Delhi: The Central Drugs Standard Control Organisation (CDSCO) has released a draft of revised Guidance for Industry document, in consultation with the stakeholders to align with the New Drugs and Clinical…
Glenmark recalls 6,528 bottles of BP drug in US
Glenmark Pharmaceuticals is recalling 6,528 bottles of Diltiazem Hydrochloride extended-release capsules, a medication used to treat high blood pressure in the American market due to failed dissolution specifications, informed USFDA…
Pharma Exports To US Surges 15% In 11 Months Of FY24
Mumbai: After a lull, India’s pharma exports to the US have surged 15% in the first 11 months of the last financial year despite regulatory challenges and rising pricing pressures. The…
‘Ram Kit’, Available At Many Drugstores In Agra
Agra: Ram Kit from Kanpur has reached Agra, it is claimed that life can be saved from heart attack in just seven rupees, know what is there in this small packet which…
Uttarakhand SLA, Ayush Ministry Stalled Action Against Patanjali: RTI Activist
New Delhi: The Uttarakhand state licensing authority did not register any case against the Ramdev-founded Patanjali Ayurveda company despite the fact that the company violated the SLA’s repeated written missives,…
CDSCO Releases Draft GDP Guidelines For Pharmaceutical Products
New Delhi: The Central Drugs Standard Control Organisation (CDSCO) has released the draft guidance document on Good Distribution Practices (GDP) for pharmaceutical products, in line with the WHO Technical Report Series…
45% Doctors In India Are Writing Incomplete Prescriptions: ICMR Survey Report
New Delhi: Around 10 percent of prescriptions from tertiary care and teaching hospitals analysed as part of a year-long government study had “unacceptable deviations” such as inappropriate prescription of of…
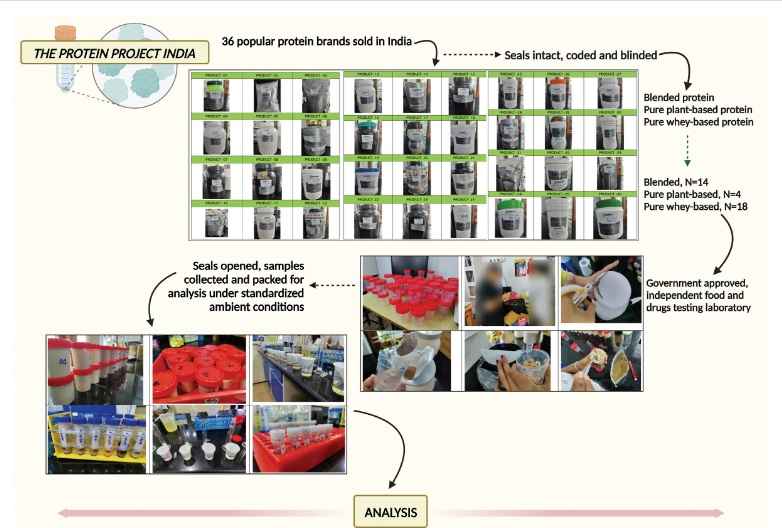
70% Of 36 Popular Protein Supplements Sold In India Mislabeled, 14% Contain Toxins, Says New Study
New Delhi: A first-of-its-kind observational analysis of the most popular protein powders sold and consumed in India has shown that the majority of these supplements falter on quality, labelling or advertised…
Notices Sent To Patanjali, But Not Under Key 1954 Law
New Delhi: Uttarkhand’s drug controller sought responses from Patanjali Ayurved over its misleading ads promising miracle cures for serious ailments under a section of the Drugs and Cosmetic Rules Act, 1945…
Generic Drug Makers Must Close ‘Knowledge Gap’ To Overcome Doctors’ Brand Loyalty: GlobalData
London: Generic drugs feature the exact same active ingredient, strength and dosing regimen as their brand-name predecessors, but, if it weren’t for their typically lower prices, they still wouldn’t be many…