Mumbai : The US drug regulator has stepped up its inspection exercise in India, and plans to have extra surprise or ‘quick discover’ inspections, ending the two-year reprieve from ‘warning letters’ and different regulatory measures that Indian drug makers have loved because the onset of the Covid-19 pandemic.
“In terms of (inspection) activity (in India) we are getting closer to pre-pandemic levels,” Sarah McMullen, nation director – India at US Food and Drug Administration (USFDA) instructed ET, on the sidelines of a current business occasion in Mumbai.
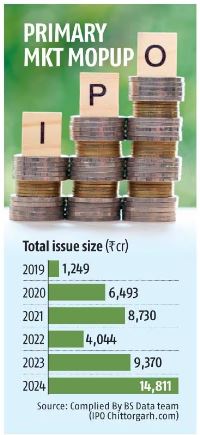
India has the biggest variety of USFDA-registered drug manufacturing services exterior of the US. The US accounted for 29% of the entire pharma exports of India woth $24.62 billion in FY22.
On a median through the pre-pandemic interval, India used to see about 200 inspections yearly. This quantity dropped to 80 in 2020, and to only 5 in 2021 attributable to Covid associated disruptions.
This resulted in official motion indications (OAIs) and warning letters falling considerably. In 2020, Indian firms obtained 25 warning letters. This quantity dropped to 2 in 2021 and stands at 3 far in 2022.
The OAIs, which ranged wherever from 40-50 a 12 months in India , too fell steeply in the pandemic interval. According to a Motilal Oswal report, Indian manufacturing websites have obtained 60 OAI citations over Sep’19-Sep’22.
The challenge of each warning letters and OAI outcomes in lack of approval on the market of recent generic medicine to the US from the involved plant, typically ensuing in a fall in the inventory value of the impacted firm. Postponed inspections additionally means delay of recent product approvals hurting the US enterprise.
USFDA needed to postpone international inspections or prioritise them primarily based on mission crucial standing for a lot of the final two years attributable to Covid pandemic. With the pandemic receding, the US company plans to have a whole lot of unannounced or quick discover inspections.