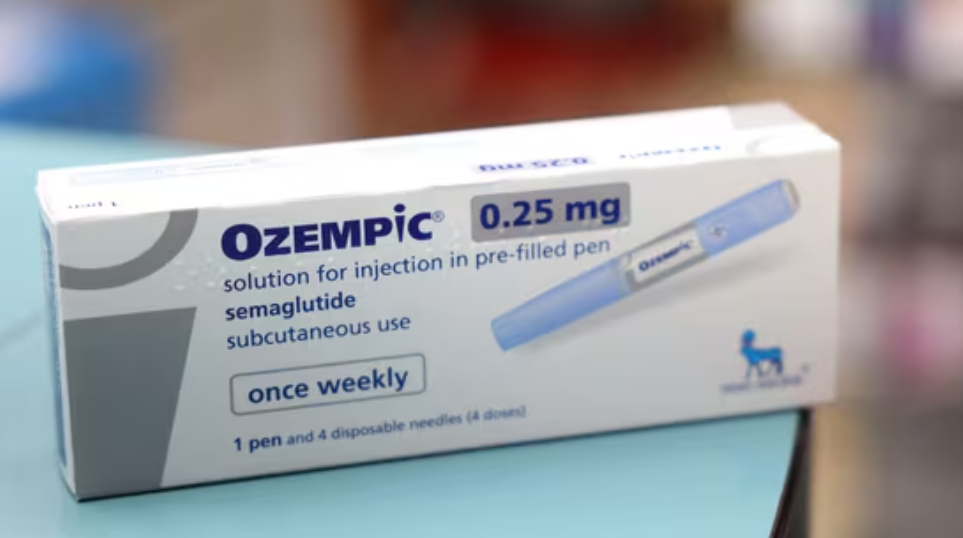
The prices of Ozempic in India can potentially halve to ₹3,600 after Dr. Reddy’s and its ilk start producing the generic version of semaglutide, Nomura says.
Dr Reddy’s Laboratories Ltd. is planning to launch a generic version of Ozempic in March after Novo Nordisk AS’s patent for the drug expires. That would make the Indian drugmaker one of the first firms to capture the gold rush for the weight-loss drug in the diabetes capital of the world.
The Hyderabad-based pharma company will sell the generic Ozempic at “competitive pricing” to ensure affordability, M.V. Ramana, its chief executive for Branded Markets, told reporters on Wednesday. The pricing will also depend on how peers price their product.
“This would be a major opportunity,” Ramana said.
Dr Reddy’s launch of generic Ozempic, used to treat Type-2 Diabetes, marks the onset of an influx of global competition that Novo Nordisk is expecting for its $30-billion franchise. Dozens of manufacturers in India and other parts of the world have geared up to sell unbranded versions for populations that previously had limited access to the blockbuster drug.
Generic semaglutide—the key ingredient in Novo Nordisk’s Ozempic and Wegovy—could be priced as low as $40 ( ₹3,600) in 2026, according to Nomura analyst Saion Mukherjee, less than half the current price of Ozempic in India.
Dr Reddy’s is preparing to manufacture 12 million pens in the first year of sales, Chief Executive Officer Erez Israeli said, adding that it will be making the active ingredient in the drug. It will also partner with other companies in India to market the drug.
The patent in India expires on 21 March, making it a key market in the firm’s strategy. Other key markets are Turkey, Brazil and Canada, he said.
Dr Reddy’s has received marketing approval and manufacturing approval from the Drugs Controller General of India for generic Ozempic, while it is awaiting a marketing nod for its non-branded version of Wegovy, Novo Nordisk’s weight-loss drug.
The company is awaiting further guidance from Health Canada after it filed a response in November to a notice of non-compliance, which showed the drug doesn’t meet regulatory requirements.
The regulator has 180 days to respond, and Dr Reddy’s expects a response by May, Israeli said, adding that they are prepared to launch immediately. “If we get approval in this timeframe, we will be first to launch in Canada.”






