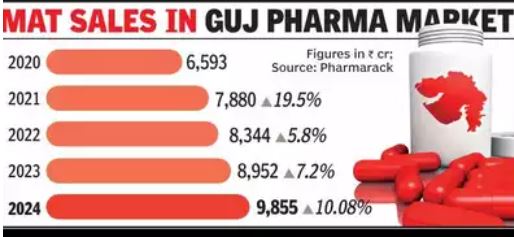
Ahmedabad: Gujarat’s pharmaceutical sector is back in the spotlight, ending 2024 on a high note with double-digit growth for the first time in three years. With Rs 9,855 crore in moving annual total (MAT) sales — 10.08% increase from Rs 8,952 crore in Dec 2023, the industry has not only bounced back from post-pandemic challenges but has also aligned itself with the national growth trajectory.
The sector’s rebound has been driven by several factors, including enhanced healthcare investments, increased demand for pharmaceutical products and a revival in export activities. Sheetal Sapale, vice-president (commercial), Pharmarack, said, “The growth is pretty much in line with the India growth, which grew at 8% during the year. Cardiac, anti-infectives and anti-diabetic therapies accounted for the maximum sales in India.”
New product launches and price factors are among other reasons driving the market’s growth. Viranchi Shah, national president of the Indian Drug Manufacturers’ Association (IDMA), said, “The recovery in Gujarat’s pharmaceutical market is a positive sign for the industry. One of the key factors has been the gradual return of post-Covid patients to clinics, contributing to an improvement in patient count. Increased govt initiatives, such as broader insurance coverage, have also played a vital role in driving accessibility and growth.”
He added, “The rise in sales volumes of off-patented drugs, coupled with the growing market for cardiovascular, metabolic and cancer therapies, highlights shifting incidences of ailments and healthcare priorities. New product launches and value growth from price adjustments are further bolstering the sector. Looking ahead, the double-digit growth trend seems set to continue, underscoring a promising outlook for the pharmaceutical industry.”






