PATNA: Bihar is currently facing a severe shortage of doctors and paramedical staff. To make matters worse, a majority of blood banks in the state are operating without “valid licences,” as highlighted in a CAG report presented in the assembly on the concluding day of the winter session on Friday.
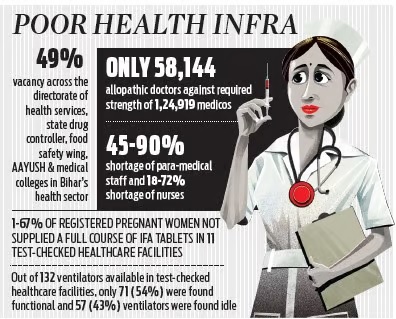
In Bihar, 1,24,919 doctors were required to serve the projected population of 12.49 crore (March, 2022) and also to fulfil the recommendation of the WHO. Against it, only 58,144 allopathic doctors were available in the state (as of January 2022), which was 53% less than the recommended norms of WHO and 32% less than the national average, said the CAG’s performance audit report on Public Health Infrastructure and Management of Health Services for the period 2016-22.
In primary and secondary healthcare facilities, 23,475 (61) and 18,909 posts (56%) were lying vacant against the total sanctioned strength.
Similarly, 49% and 82% posts were lying vacant against sanctioned strength in tertiary and AYUSH healthcare facilities, respectively. There was an overall shortage of 35,317 (60%) against the sanctioned strength, the CAG report added.
In connection with the blood banks, the report said, “Audit checked the record of six blood banks and critical shortage of equipment was observed during inspections, hence their licences were not renewed. However, blood banks continued to operate with such critical shortages, without being in possession of valid licences.”
The report said scrutiny of the purchase orders data of the Bihar Medical Services and Infrastructure Corporation Ltd, a nodal agency of the state government for procuring and managing drugs for all medical institutions, revealed that from 2016-17 to 2021-22, supplies of the medicines were received with only 35% to 74% of their shelf life. In certain cases drugs nearing expiry were not withdrawn,” it said.
Physical verification of 25 ambulances showed that none of the ambulances had required equipment, medicine or consumables.






