Southern Denmark: In their separate studies, Jakob Grauslund, Professor of Ophthalmology, and Anton Pottegård, Professor of Pharmaceuticals, have used two different methods to investigate all Danish users of Ozempic.
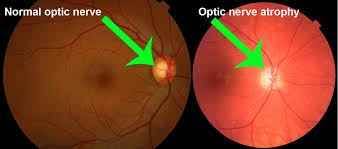
Both of the new register-based studies from SDU confirm a concern that was raised earlier this year in a smaller American study. In the American study, it was observed that Ozempic more than doubles the risk of the rare condition NAION (non-arteritic anterior ischemic optic neuropathy), which causes damage to the optic nerve of the eye.
Jakob Grauslund and his research colleagues have validated the hypothesis of the American study in Denmark, where the drug Ozempic is used by 106,454 patients in the treatment of type 2 diabetes.
– We have examined data from all 424,152 Danes with type 2 diabetes. We found that Ozempic more than doubles the risk of developing NAION. NAION is damage to the optic nerve of the eye caused by a sudden stop of blood flow to the optic nerve. It is a condition that can lead to severe and permanent loss of vision, says Jakob Grauslund, Professor of Ophthalmology at the Department of Clinical Research, SDU, and Department of Ophthalmology E, Odense University Hospital.
– In the study, we found that the risk is doubled. At the same time, in the clinic we have noticed that since Ozempic came on the market in 2018, the number of NAION cases in Denmark has increased, explains Jakob Grauslund.
– Whereas we used to see between 60 and 70 cases of NAION per year, we are now seeing up to 150. In addition, we have noticed that the patients who come to hospital due to NAION are more often patients with type 2 diabetes.
In Jakob Grauslund’s study, the researchers divided the patients into two groups: those who were treated with Ozempic and those who received other treatment. In the statistical analyses, age, gender, blood sugar and a wide range of other conditions were also taken into account.






