Hyderabad: The Drugs Control Administration (DCA) of Telangana has intensified its efforts against illegal drug practices across the state, conducting multiple raids to curb violations. The actions included the seizure of unlicensed medicines, overpriced drugs, misleading advertisements, and falsified nutraceutical products.
1. Raid on Unlicensed Medical Shop in Adilabad
DCA officials raided a medical shop in Wankidi Village, Neradigonda Mandal, Adilabad District, operating without a valid drug license. The shop, run by Abdul Rahim, was found stocking 31 varieties of medicines, including antibiotics, analgesics, and steroids, worth ₹15,000.
Samples have been collected for analysis, and legal action will follow. Wholesalers supplying medicines to unlicensed entities have also been warned of strict penalties under the Drugs and Cosmetics Act.
2. Action Against Quack’s Clinic in Jubilee Hills
In Hyderabad, DCA officials raided the clinic of Syed Azam, an unqualified practitioner operating in Shaikpet, Jubilee Hills. The clinic stocked 22 varieties of medicines, including high-generation antibiotics and steroids, valued at ₹10,000. Officials highlighted the public health risks posed by unqualified practitioners, including antimicrobial resistance and severe side effects from the misuse of steroids.
3. Overpricing of Essential Medicines
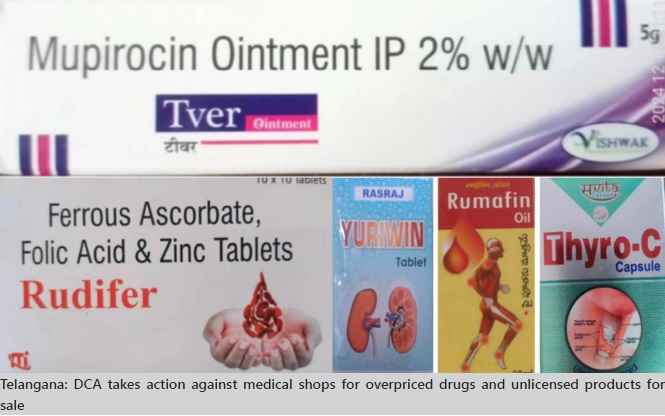
DCA seized two medicines being sold at prices far above the ceiling rates fixed by the Central Government:
• Tver Ointment (Mupirocin Ointment IP 2%): Sold at ₹135 for 5 gm, exceeding the permissible price of ₹113.62.
• Sazolet 500 mg (Sulfasalazine Tablets): Sold at ₹65.50 for ten tablets, surpassing the approved ceiling price of ₹52.86.
The products were seized in Sangareddy District, and investigations are underway. Officials reiterated that overpricing of essential medicines is a punishable offense under the Drugs (Prices Control) Order, 2013.
4. Seizure of Falsified Nutraceutical Products
In Hayathnagar, Ranga Reddy District, officials detected Rudifer Tablets, falsely marketed as a food product under an FSSAI license. Manufactured by Maxtra Laboratories, the product failed to comply with the Drugs and Cosmetics Act, 1940. Such practices endanger public health, as these drugs often do not meet prescribed manufacturing and quality standards.
5. Misleading Advertisements for Drugs
The DCA identified several products with false medical claims, violating the Drugs and Magic Remedies (Objectionable Advertisements) Act, 1954.
Officials emphasized that misleading advertisements can harm public health and are prohibited by law.






