New Delhi: The World Health Organization (WHO) has sought clarification from India on whether the cough syrup linked to over 15 child deaths in the country has been exported to other countries, a senior official of the global health agency said on Wednesday (October 8, 2025).
The WHO is yet to issue a Global Medical Products Alert on Coldrif syrup, the cough syrup which has allegedly caused the child deaths in Madhya Pradesh and Rajasthan. The official added that the need for an alert will be investigated only after receiving a response from Indian health authorities.
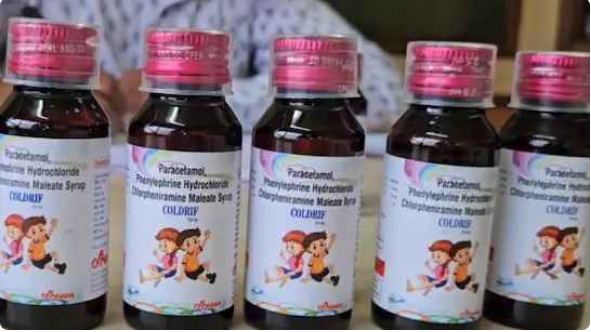
So far, at least 17 children aged below five have died in India, allegedly after consuming cough syrup containing a toxic compound diethylene glycol (DEG). Coldrif was manufactured by Sresan Pharmaceuticals, based in Tamil Nadu. The company is currently under investigation.
Exports of cough syrups from India have been mandated to clear additional testing at government-mandated laboratories since 2023 following the deaths of over 140 children in the Gambia, Uzbekistan and Cameroon. The deaths were linked to syrups made in India.
The Central government, in an order dated December 18, 2023, had said that the fixed-dose combination (FDC) of chlorpheniramine maleate IP 2mg and phenylephrine HCl IP 5mg drop/ml “should not be used in children below four years of age.” Prescribed to treat symptoms of cold and cough, including runny nose, sneezing and sore throat and watery eyes, Coldrif contains chlorpheniramine maleate, paracetamol and phenylephrine.
The order, issued by the Central Drugs Standard Control Organisation, and signed by Rajeev Singh Raghuvanshi, Drugs Controller General of India, ordered firms to “mention warning” in this regard on the label and the package insert.






