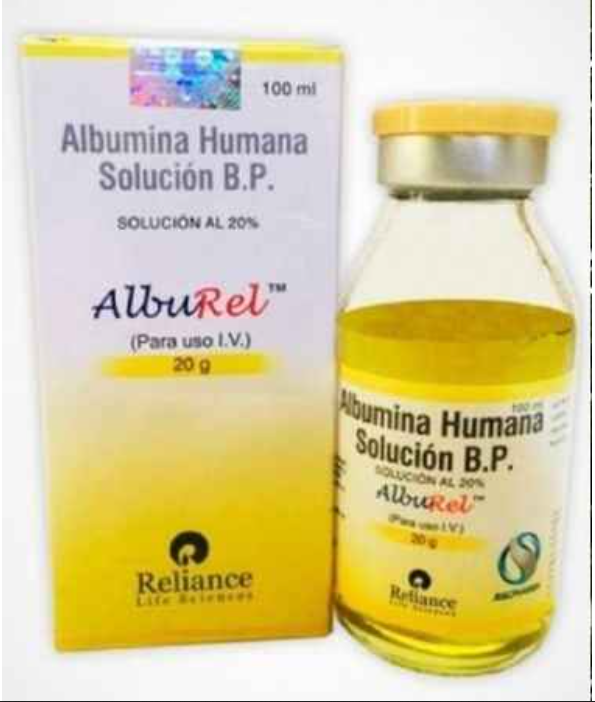
Pune— In a major breakthrough exposing vulnerabilities in India’s pharmaceutical supply chain, the Food and Drug Administration (FDA) Pune Division has declared batches of “RituxiRel” (a counterfeit version of Rituximab, used for cancer and autoimmune diseases) and “Alburel” (Human Normal Albumin IP, critical for treating liver conditions, burns, and shock) as spurious, leading to an FIR against multiple firms across Maharashtra. The racket, uncovered through routine inspections and lab testing, highlights how fake life-saving injections are infiltrating markets, potentially endangering thousands of patients. This follows earlier seizures in September-October 2025 worth over ₹4.5 lakh, with links now traced to Delhi and Chandigarh, underscoring an organized interstate operation.
The Bust: Seizures and Declarations
The investigation began on July 28, 2025, when FDA inspectors collected samples of RituxiRel and Alburel during a routine check at Maitri Pharmaceuticals in Sadashiv Peth, Pune. The Alburel batch (No. AN20G25084) was sent for testing to the Government Analyst in Mumbai, who declared it “spurious” on February 2, 2026, after it failed the Indian Pharmacopoeia (IP) identification test under Section 17-B of the Drugs and Cosmetics Act, 1940. The counterfeit drugs bore fake labels claiming manufacture by Reliance Life Sciences Nashik Pvt. Ltd., which denied producing the batch in a February 5, 2026, response, providing comparison data on packaging discrepancies.
Earlier in September 2025, a joint FDA-Pune City Police raid targeted Shriram Healthcare in Gangadham Chowk, Pune, seizing 39 bottles of fake Alburel valued at ₹3.41 lakh. This led to further raids in Mumbai and Raigad, uncovering a total stock worth ₹4.5 lakh. By October 2025, the racket was described as spanning multiple cities, with counterfeit injections posing “serious health risks.”
Involved Parties and Supply Chain
The spurious injections were distributed through a complex network:
Maitri Pharmaceuticals (Pune): Procured stock from Shri Sai Logistics (Nagpur) on July 22, 2025.
Shri Sai Logistics (Nagpur): Sourced from Nexus Lifecare Pvt. Ltd. (Mumbai).
Nexus Lifecare Pvt. Ltd. and NRX Distributors LLP (Mumbai): Received consignments from RRT Pharma (Kamothe, Raigad) via invoices in July-August 2025.
RRT Pharma (Raigad): Owner Ritesh Rohidas Thombare (also spelled Thombre in some reports) failed to provide purchase/sale records, claiming via email they were unavailable.
In the September 2025 bust, Shriram Healthcare owner Shriram Nandkishor Chandak sourced from R.S.L. Pharma (Sadashiv Peth, Pune; partners Rajesh Shantaram Jog and Shailam Gatla), which linked back to N.R.A.X. Pharma (Sion, Mumbai) and R.R.T. Pharma. Arrests included Mohd. Danish Khan (Delhi resident), with ties to Chandigarh suppliers. FIRs were filed at Market Yard and Vishrambaug Police Stations, with Police Inspector Arun Dattu Ghodke leading the probe.
Legal Actions and Charges
The latest FIR at Vishrambaug Police Station invokes Sections 318(4) (cheating), 336(3) (endangering life), 340(2) (wrongful confinement), 278 (fouling atmosphere), 60 (attempt to commit offence), and 3(5) (criminal conspiracy) of the Bharatiya Nyaya Sanhita (BNS) 2023. An earlier complaint at Market Yard targeted RRT Pharma’s owner under similar IPC provisions. FDA issued notices for recalls and record submissions.
Health Risks and Public Impact
These counterfeit injections pose grave dangers: Alburel is vital for plasma volume expansion in critical care, while RituxiRel mimics Rituximab for treating lymphoma, rheumatoid arthritis, and other conditions. Fake versions could contain incorrect or harmful substances, leading to treatment failure, allergic reactions, or death. Authorities noted the racket defrauds patients and the government, with the accused conspiring to pass off fakes as genuine.
Official Statements and Ongoing Probe
FDA Joint Commissioner Girish Hukre stated: “The investigation is now focused on tracing the source of the counterfeit stock and identifying whether the drug was manufactured locally or brought from outside. Police have registered an offence… and further action is underway.” Drug Inspector Shrutika Kamlaring Jadhav filed the complaint, emphasizing enforcement under the Drugs and Cosmetics Act.
This case echoes broader concerns over counterfeit drugs in Maharashtra, with similar rackets busted for Chymoral Forte (linked to Bihar) and other medicines. Health experts urge patients to verify drug authenticity via QR codes or apps and report suspicions. The government faces calls for stricter supply chain monitoring to prevent such threats to public health.






