Six Women Develop Serious Complications After C-Section
Jaipur/Kota — In a serious medical incident that has raised concerns over patient safety at government facilities, six women who underwent caesarean section deliveries at Kota’s New Medical College Hospital developed…
Pak-made illegal cosmetics recovered from Itwari shop
Nagpur: Maharashtra Food and Drug Administration (FDA), Nagpur Division, raided a cosmetics firm in Itwari market and seized Pakistan-origin beauty products suspected to have harmful mercury and steroids last week. The…
Stem cell therapy for autism illegal: NMC advisory
New Delhi: The National Medical Commission (NMC) issued a strict advisory on March 25, 2026, declaring stem cell therapy illegal for treating Autism Spectrum Disorder and cerebral palsy in routine…
NPPA allows 0.64% hike in MRP of essential drugs in line with WPI rise
New Delhi: The National Pharmaceutical Pricing Authority (NPPA) on Wednesday announced that manufacturers may increase prices of scheduled formulations included in the National List of Essential Medicines (NLEM) by 0.64…
Study finds rising resistance to a last-resort antibiotic in Africa
Ethiopia: Resistance to a last-resort antibiotic is rising sharply in Africa to two multidrug-resistant (MDR) bacterial pathogens that pose major threats in health care settings, according to a study this week…
No spurious drugs were detected in the state during the 2024–25 financial year and so far in 2025–26: CM Manik Saha in Assembly
Agartala: Chief Minister Prof. (Dr.) Manik Saha on Tuesday informed the Tripura Legislative Assembly that the State Drugs Testing Laboratory (SDTL) has initiated action against 62 cases of “not of…
India’s Medical Devices Sector Logs 4,108 Licensed Manufacturers and ₹20,658 Crore FDI Inflows Since 2018
New Delhi— The government has reported robust growth in India’s medical devices industry, with 4,108 licensed manufacturers currently operating across the country and cumulative Foreign Direct Investment (FDI) inflows touching…
Abbott introduces its most advanced drug eluting stent XIENCE Skypoint™ in India
NEW DELHI— Abbott, the global healthcare company, today announced the launch of XIENCE Skypoint in India. This is Abbott’s most advanced generation stent within the globally trusted XIENCE™ family of…
DCGI asks drugmakers to stop GLP-1 obesity awareness ad campaigns
New Delhi: The Central Drugs Standard Control Organisation (CDSCO) has asked all drugmakers having marketing authorisation to sell glucagon-like peptide-1 (GLP-1) agonist drugs for obesity management to not partake in…
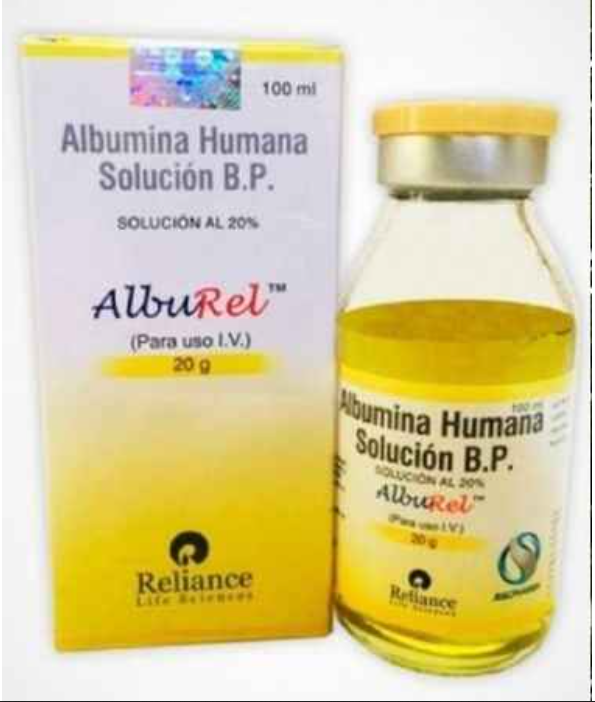
Multi-State Supply Chain of Fake RituxiRel and Alburel Injections Exposed Amid Health Risks
Pune— In a major breakthrough exposing vulnerabilities in India’s pharmaceutical supply chain, the Food and Drug Administration (FDA) Pune Division has declared batches of “RituxiRel” (a counterfeit version of Rituximab,…