A total of 44 US states on Monday filed a lawsuit against as many as 20 generic drug manufacturers, including seven Indian firms. Among these Indian firms, 5 have been sued by the state attorney generals and 4 are under DOJ (Department of Justice) investigation.
The multi-state lawsuit seeks damages and civil penalties against the pharma companies for allegedly running a coordinated campaign to artificially inflate the prices of more than 100 drugs between 2013 and 2015.
How will the drug firms be penalised?
According to a research report by JM Financial, the investigation against these pharma companies could go on for several years, but they could still face a lot of damage due to the lawsuit. The fines can run into millions of dollars.
 “Under the Sherman anti-trust Act, a maximum penalty of USD 100 million could be imposed. However, the maximum penalty can be increased to a fraction (c.20%) of the volume of affected commerce during the conspiracy period, if the amount is greater than USD 100 million,” the report said.
“Under the Sherman anti-trust Act, a maximum penalty of USD 100 million could be imposed. However, the maximum penalty can be increased to a fraction (c.20%) of the volume of affected commerce during the conspiracy period, if the amount is greater than USD 100 million,” the report said.
There can be severe penalties levied on the Indian pharma companies too. Glenmark could face the highest penalty of $873 million, while Aurobindo the least at $13 million. Glenmark has said they “expect to file papers with the Federal Court in due course denying the accusations”.
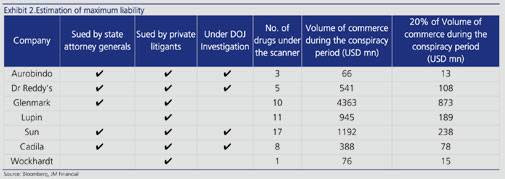
Taro Pharmaceutical, a subsidiary of Sun Pharma, which is India’s largest pharmaceutical company by sales could face a liability of $238 million, as per the JM Financial’s report. It also said that: “Firms named in one or few drugs (Aurobindo & Wockhardt) might consider settling earlier to avoid high defence costs and the risk of shared liability.”
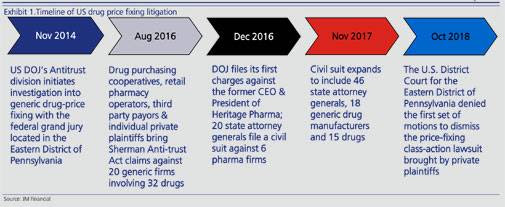
Timeline of the 5 year long investigation:
The lawsuit accuses Teva Pharmaceuticals USA Inc of orchestrating a sweeping scheme with 19 other companies to inflate drug prices. In case of certain drugs the prices were increased by more than 1,000%. During a 19-month period from 2013 to 2015, Teva significantly raised prices of about 112 generic drugs and colluded with its competitors on at least 86 medicines, the states claimed.
The 7 Indian drug makers named in the lawsuit are Wockhardt, Dr Reddy’s Laboratories, Aurobindo Pharma, Glenmark Pharmaceuticals, Lupin, Zydus Pharma (Cadila Healthcare), and Taro Pharmaceutical Industries – a subsidiary of Sun Pharma, which is India’s largest pharma company by sales. Reacting to the news, Indian pharma stocks took a beating on Monday. BSE’s healthcare index fell 3.53% to 13,310.47 points. Shares of Sun Pharmaceuticals plunged 21% in intraday trading, but ended the day down 9.39% to Rs 396.85 while Dr Reddy’s fell 2.5% to Rs 2,804.95. However, the sector is in the green today, trading 86 points higher at 13,396 level.






