CDSCO to dispose online applications pending for more than two years
New Delhi: In an effort to reduce the backlog and clear the long-pending applications with lack of response from the applicants, the Central Drugs Standard Control Organisation (CDSCO) has introduced…
DoP may exempt 11 more drugs from public procurement rules
New Delhi: The Department of Pharmaceuticals (DoP) has proposed to add 11 more drugs including insulin and monoclonal antibodies to the list of 84 drug formulations it has mentioned in…
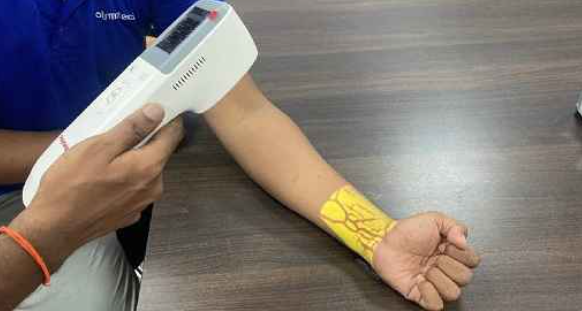
Polymatech begins domestic manufacturing of vein finder devices
CHENNAI: Polymatech Electronics Limited today announced a landmark milestone in India’s medical device sector with the successful registration and commencement of domestic manufacturing of its Vein Finder devices in accordance…
DoP includes member of HTAIn in expert committee on pricing of drugs
New Delhi: Department of Pharmaceuticals (DoP) has added an expert representative from the Health Technology Assessment of India (HTAIn) under the Department of Health Research (DHR) as a member of…
US FDA Warns Two Drugmakers Including Medgel From India Over DEG, EG Testing Lapses
Maryland : The US Food and Drug Administration (FDA) has reprimanded two drugmakers — Avlon Industries based in Melrose Park, Illinois, and Medgel based in Pithampur, Madhya Pradesh, India — over…
DCGI Asks Manufacturers Not To Submit Same Batch Of Cough Syrup To More Than One Lab
New Delhi : The Drugs Controller General of India (DCGI) has directed the manufacturers not to submit the same batch of cough syrup to more than one laboratory at a time,…