Centre gives custom cushion to supply strained Pharma, MedTech manufacturers
New Delhi: In an effort to curb the incremental cost burden arising from the in West Asia, the Centre has announced customs duty exemptions on 40 petrochemical inputs, providing relief…
Raj HC quashes license cancellation of medical agency for RGHS irregularities
Jodhpur: The Rajasthan High Court has quashed an order cancelling the drug license of a Jodhpur-based medical agency, holding that the Licensing Authority cannot impose the “extreme penalty” of cancellation…
Abbott introduces its most advanced drug eluting stent XIENCE Skypoint™ in India
NEW DELHI— Abbott, the global healthcare company, today announced the launch of XIENCE Skypoint in India. This is Abbott’s most advanced generation stent within the globally trusted XIENCE™ family of…
Top 30 pharma companies post 12% revenue growth in first nine months
Mumbai: The aggregate financial performance of top 30 Indian pharmaceutical companies, with revenues of over Rs. 2,500 crore during 2024-25, remained under pressure during the first nine months of FY2025-26…
DCGI asks drugmakers to stop GLP-1 obesity awareness ad campaigns
New Delhi: The Central Drugs Standard Control Organisation (CDSCO) has asked all drugmakers having marketing authorisation to sell glucagon-like peptide-1 (GLP-1) agonist drugs for obesity management to not partake in…
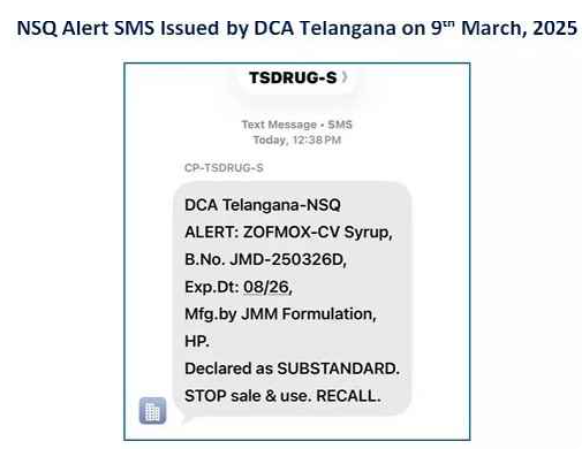
Telangana DCA launches SMS alert system to notify NSQ drugs
Hyderabad: The Drugs Control Administration (DCA) in Telangana has significantly intensified its crackdown on illegal medical practices while simultaneously launching a high-tech digital shield for public health. On March 9, the…
Centre of Excellence in Nutraceuticals inaugurated by the Hon’ble Chief Minister of Kerala
T’puram: Chief minister Pinarayi Vijayan virtually inaugurated a centre of excellence in nutraceuticals (CoEN) at Bio 360 Life Sciences Park, Thonnakkal, on Monday. The initiative, spearheaded by Kerala State Council…
NSQ reporting nearly doubles in 2025, buoyed by greater state participation
New Delhi: The number of drug samples declared as Not of Standard Quality (NSQ) by the drug regulators in the country has registered over two-fold growth during the 12 months of…
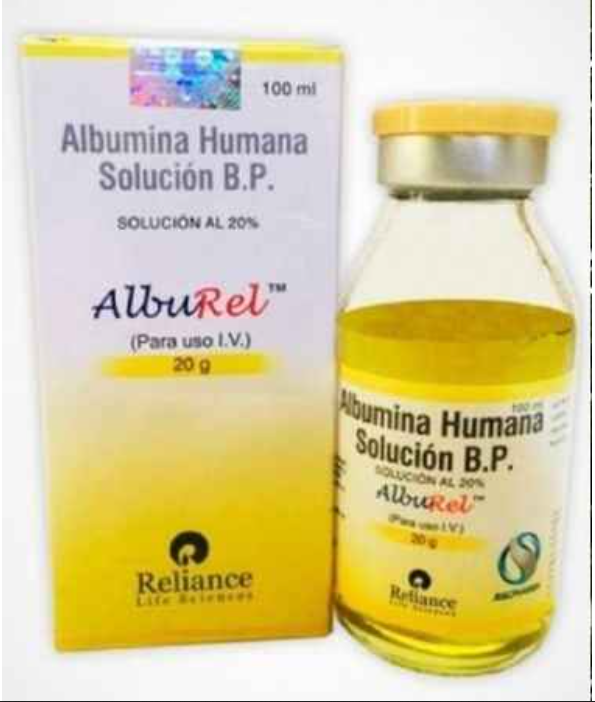
Multi-State Supply Chain of Fake RituxiRel and Alburel Injections Exposed Amid Health Risks
Pune— In a major breakthrough exposing vulnerabilities in India’s pharmaceutical supply chain, the Food and Drug Administration (FDA) Pune Division has declared batches of “RituxiRel” (a counterfeit version of Rituximab,…
TN, Karnataka, Rajasthan declare highest number of NSQs in January
New Delhi: Around 29% of the total Not of Standard Quality (NSQ) drug samples reported by the State drug testing laboratories in the month of January were collected and tested…