Delhi Police bust fake medicine packaging racket; two arrested
New Delhi: The Cyber Cell of the Delhi Police’s Crime Branch has busted a racket involved in the illegal printing of packaging material for spurious medicines and fake cosmetic products,…
PlasmaGen Biosciences secures ₹150 crore to expand into global markets
Biopharmaceutical company PlasmaGen Biosciences raised ₹150 crore in a new round of equity financing, valuing the company at over ₹1,500 crore. ViNS Bioproducts, a specialty biopharma company, picked up a…
Cipla launches inhalable insulin for diabetes care
New Delhi, Drug firm Cipla on Monday said it has launched orally inhaled insulin powder for diabetes patients in the country. The company had obtained regulatory approval from the Central…
Indian ‘Mantra’ for USA to make Robotic Surgery Affordable and Accessible there
MedTech Startup Files FDA 510(k) for Homegrown Surgical Robot SSIMantra New Delhi: Indian medtech company SS Innovations, maker of surgical robot SSIMantra has moved ahead to become emblem of affordability…
Centre draws a price line to curb cheap pharma imports
New Delhi: The Indian government has set a minimum import price (MIP) for certain key pharmaceutical inputs to combat aggressive undercutting and dumping from Chinese manufacturers. The government imposed the…
47 Himachal-made medicines fail quality tests, companies asked to recall stock
These samples were part of a larger batch of 200 drugs tested nationwide that were found to be below standard, prompting the Central Drugs Standard Control Organisation (CDSCO) to issue…
Swell of Footfalls for Respiratory & Cardiac Emergencies in Aakash Healthcare due to Pollution
Experts of the Multispecialty Hospital warn of the Consequences for Children and Elderly New Delhi: Pollution woes in Delhi are piling up and showing in hospitals. Swell of footfalls in…
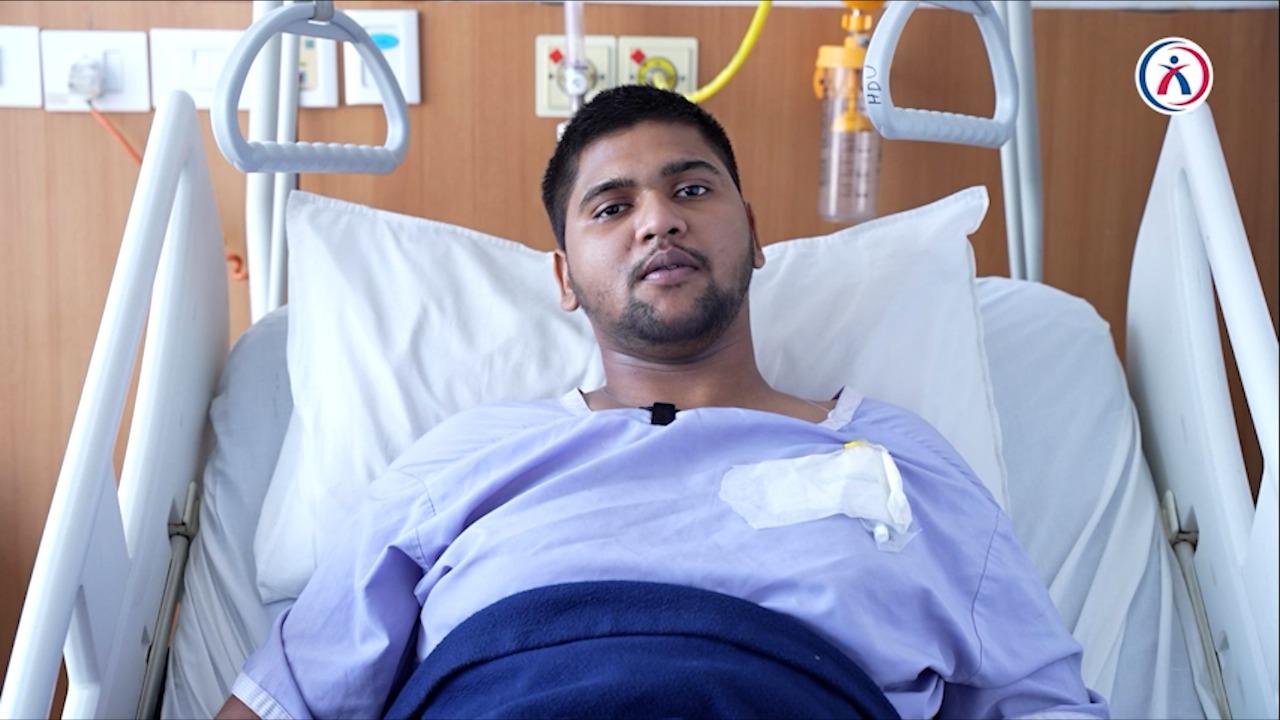
Early Morning Drowsy Road Drivers Beware! Maybe headed to ‘Air Crash’
Aakash Healthcare averted long term Complications from ‘Aviator’s Fracture’ New Delhi: Sleep deprivation is making both pilots and drivers on the road prone to ‘crash’. 21 year old sleep starved…
Granules India arm gets 5 USFDA observations for Hyderabad facility
Telangana: Granules India has announced that the U.S. Food and Drug Administration (FDA) has concluded an inspection with five observations at the Hyderabad facility of Granules Life Sciences, a wholly…
Drug regulator CDSCO flags 205 drug samples as not of standard quality in November
Central and state drug testing laboratories identified substandard and spurious medicines during routine nationwide surveillance India’s apex drug regulator, the Central Drugs Standard Control Organisation (CDSCO), has flagged 205 drug…