CDSCO to dispose online applications pending for more than two years
New Delhi: In an effort to reduce the backlog and clear the long-pending applications with lack of response from the applicants, the Central Drugs Standard Control Organisation (CDSCO) has introduced…
Brazil, Nigeria emerge as key export destinations; market expands to $20.48 billion
New Delhi: Even as global economic uncertainty drags down trade, Indian pharmaceutical exporters are seeing growth in Brazil and Nigeria as both countries are emerging as important overseas markets. Data…
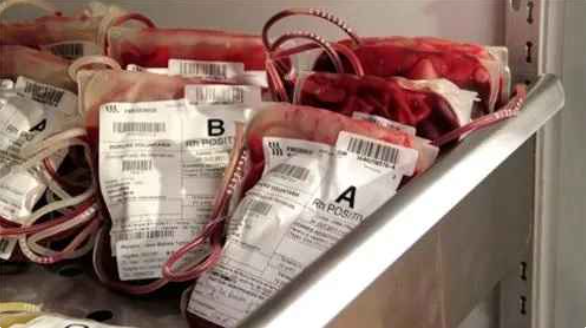
Centre flags serious lapses in state’s blood banks
Jaipur: Union health ministry has flagged “serious lapses” in Rajasthan’s blood bank management after inspections found shortcomings including inadequate record-keeping, lack of proper ELISA testing, and failures to communicate HIV-positive…
Unlicensed medical device stock worth Rs. 4.47 lakh seized in Gurugram raid
In a significant crackdown on the illegal sale of medical equipment, a team of drug control officers in Haryana conducted a high-profile raid at a private firm in Gurugram on…
Controlled substances charge: Court summons Ramdev, Amazon founder
Hisar: The court of chief judicial magistrate Gitanjaali Goel in Ambala has issued summons to 14 people, including yoga guru Baba Ramdev, Acharya Balkrishna, and Amazon founder and executive chairman…
Allahabad HC refuses to quash case against pharma company linked to cough syrup deaths in Uzbekistan
Allahabad: The Allahabad High Court recently declined to quash proceedings under the Drugs and Cosmetics Act against the directors of a company accused of manufacturing sub-standard cough syrup that caused…
IPC registers 44 small-scale medtech firms under ADRMS
Mumbai: The Indian Pharmacopoeia Commission (IPC) has registered 44 small-scale medtech firms under ADR Monitoring System (ADRMS) to help them report medical devices related serious adverse events (SAEs) for patient and…