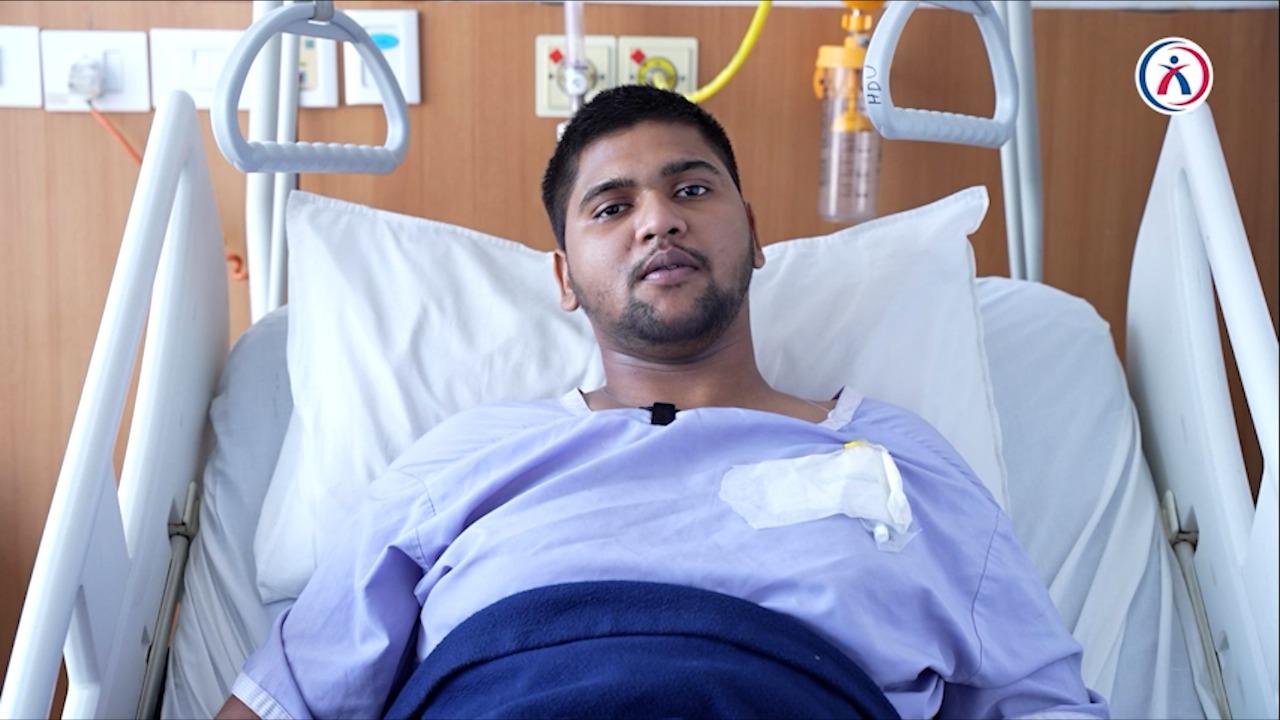
Early Morning Drowsy Road Drivers Beware! Maybe headed to ‘Air Crash’
Aakash Healthcare averted long term Complications from ‘Aviator’s Fracture’ New Delhi: Sleep deprivation is making both pilots and drivers on the road prone to ‘crash’. 21 year old sleep starved…
Granules India arm gets 5 USFDA observations for Hyderabad facility
Telangana: Granules India has announced that the U.S. Food and Drug Administration (FDA) has concluded an inspection with five observations at the Hyderabad facility of Granules Life Sciences, a wholly…
Drug regulator CDSCO flags 205 drug samples as not of standard quality in November
Central and state drug testing laboratories identified substandard and spurious medicines during routine nationwide surveillance India’s apex drug regulator, the Central Drugs Standard Control Organisation (CDSCO), has flagged 205 drug…
Trump announces historic drug price cuts; Move likely to impact Indian pharma sector
The move aims to align American drug costs with the lowest international standards, a strategy that could significantly impact major generic suppliers like India Washington: In a major policy shift…
Aurobindo Pharma subsidiary’s plant in Andhra Pradesh issued five observations by U.S. FDA
The U.S.Food and Drug Administration has issued five observations to Aurobindo Pharma subsidiary APL Healthcare’s unit in Andhra Pradesh. The U.S.FDA inspected Unit-IV of APL Healthcare in SPSR Nellore District,…
Crackdown on Illegal Ayurvedic Syrup Factory: An Inside Look
In Madhya Pradesh’s Indore district, officials sealed an illegal Ayurvedic syrup factory located in a residential area. The factory, overseen by Surendra Singh, was found lacking a quality-test lab and…
Sun Pharma Baska facility gets OAI status from USFDA
Mumbai: Sun Pharma has announced that the company has received Official Action Indicated (OAI) status from the U.S. Food and Drug Administration (USFDA) for Baska facility. The USFDA conducted an…
AstraZeneca Pharma India wins CDSCO permission to import breast cancer drug Datverzo
Bangalore: AstraZeneca Pharma India Limited has received permission from the Central Drugs Standard Control Organization (CDSCO), Directorate General of Health Services, Government of India to import for sale and distribution…
Class 8 Dropouts Used YouTube Tutorials to Make Fake Betnovate-C, Clop-G Creams, Delhi Police Bust 6-Year Racket
New Delhi: Delhi Police have busted a large-scale fake medicine manufacturing racket that had been operating for nearly six years, allegedly run by two Class 8 failures who learned to…
Samples of drugs used in diabetes, hypertension, allergies, infections found substandard in quality tests
Jaipur: Samples of five drugs were found to be of substandard quality upon testing and analysis conducted by the Commissionerate, Food Safety and Drug Control (CFSDC). On Wednesday, the CFSDC…