CDSCO Cracks Whip On Drug Manufacturing Units Over Quality Lapses
Mumbai: Around 36 per cent of the pharmaceutical manufacturing units inspected by the Indian drug regulator in recent times were forced to shut down due to non-compliance with quality standards, a…
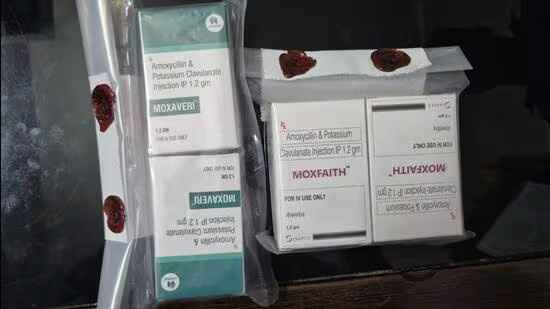
GB Nagar: Himachal Drug Makers Booked For Sub-Standard Medicines
NOIDA: The drugs control department of Gautam Budh Nagar district has registered a case against two Himachal Pradesh-located manufacturers/producers for allegedly making and selling “unfit” antibiotic medicines, officials aware of…
Glenmark Recalls 114 Batches Of Low Potassium Drug In The US Over Potentially Deadly Hyperkalemia Risk
MAHWAH, N.J. : While patients with low potassium often need medication to boost levels of the electrolyte in the body, too much of a good thing can cause serious health concerns.…
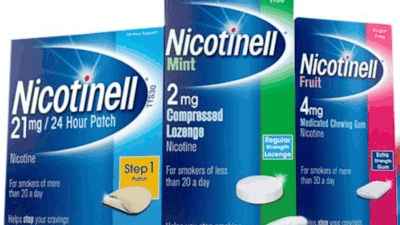
Dr Reddy’s Arm To Buy Haleon Plc’s NRT Business, Including Nicotinell
HYDERABAD: Pharma major Dr Reddy’s Laboratories is acquiring British consumer healthcare player Haleon Plc’s entire nicotine replacement therapy (NRT) business, outside of the United States market, for a consideration of GBP…
Maharashtra Drug Inspector Arrested For Taking Rs 1 Lakh Bribe In Palghar
Palghar: Maharashtra’s Palghar district witnessed a dramatic turn of events on Wednesday as the Anti-Corruption Bureau (ACB) arrested drug inspector Arti Shirish Kambli and Krishnakumar Asaram Tiwari for allegedly demanding…
Gang involved in illegal drug trade busted; 11 arrested
Varanasi: In a joint operation of SWAT Surveillance and Kotwali police, a gang involved in illegal drug trade was busted in Ghazipur on Sunday night. As many as 11 members of the gang were arrested and medicines, injections…
Spurious Drug Factory Busted In Gandhinagar
Ahmedabad: The Gujarat Food and Drug Administration (FDCA) raided a factory operating illegally within the Gujarat Industrial Development Corporation (GIDC) estate in Gandhinagar. The unit, owned by Bhavin Patel, was…
DCA Seizes Overpriced, Misleading Claim Medicine
Hyderabad: Officials from the Telangana Drugs Control Administration (DCA) in separate raids in Sangareddy and Suryapet districts seized overpriced drugs and also medicines with misleading advertisements. During a special drive, DCA…
DoP Further Expands List Of Special Invitees Into Committee Constituted To Reform Pricing Framework
New Delhi: The Department of Pharmaceuticals (DoP) has once again expanded the list of special invitees for its Committee for reforms in the pricing framework for drugs and medical devices, formed…
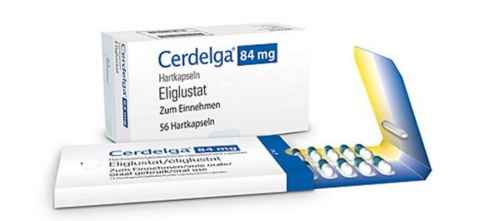
India Key Market For APIs, But Patients Forced To Cough Up Crores To Buy Orphan Drugs
HYDERABAD: Though India manufactures most of the key active pharmaceutical ingredients (APIs) for over 400 FDA-approved orphan drugs for rare diseases, patients back home are forced to cough up crores to…